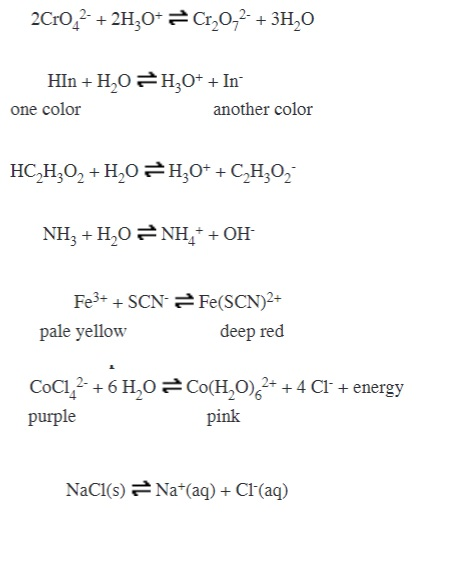![Fe(H2O)5(NO)]2+, das Chromophor des “braunen Rings” - Monsch - 2019 - Angewandte Chemie - Wiley Online Library Fe(H2O)5(NO)]2+, das Chromophor des “braunen Rings” - Monsch - 2019 - Angewandte Chemie - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/acd53743-3df9-4234-a3d9-a35cbb84a9a5/ange201902374-fig-0003-m.jpg)
Fe(H2O)5(NO)]2+, das Chromophor des “braunen Rings” - Monsch - 2019 - Angewandte Chemie - Wiley Online Library
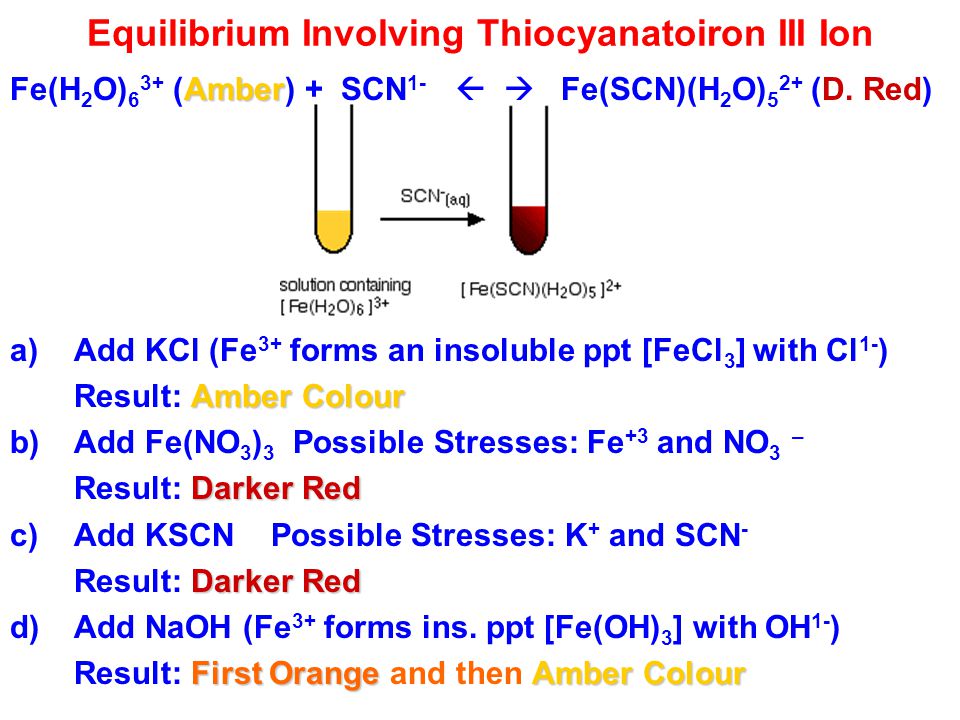
![How does the complex ion [Fe.(H20) 5.SCN] 2+ form from [Fe(H2O6)]3+ + SCN-? ←→ [Fe.(H20) 5.SCN] 2+ - Quora How does the complex ion [Fe.(H20) 5.SCN] 2+ form from [Fe(H2O6)]3+ + SCN-? ←→ [Fe.(H20) 5.SCN] 2+ - Quora](https://qph.cf2.quoracdn.net/main-qimg-697ede5c75c8b3a70ee9cb370c4d8bf9.webp)
How does the complex ion [Fe.(H20) 5.SCN] 2+ form from [Fe(H2O6)]3+ + SCN-? ←→ [Fe.(H20) 5.SCN] 2+ - Quora
How does the complex ion [Fe.(H20) 5.SCN] 2+ form from [Fe(H2O6)]3+ + SCN-? ←→ [Fe.(H20) 5.SCN] 2+ - Quora
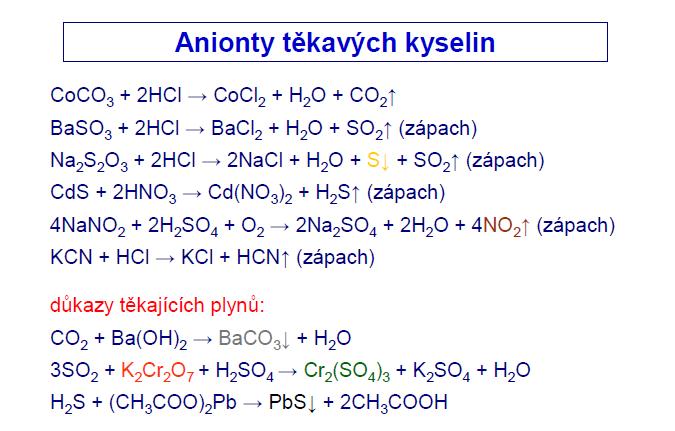
Chem. Listy 102, 599-609 (2008) Sekce 1 - přednáłky 599 1L-01 CHEMIE KARBENOVĐCH KOMPLEXŮ CHROMU A ŠELEZA DALIMIL DVO
![Fe(H2O)5(NO)]2+, das Chromophor des “braunen Rings” - Monsch - 2019 - Angewandte Chemie - Wiley Online Library Fe(H2O)5(NO)]2+, das Chromophor des “braunen Rings” - Monsch - 2019 - Angewandte Chemie - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/cf68558a-6e5b-4f05-96e9-61ce3082279a/ange201902374-toc-0001-m.jpg?trick=1669415101026)



![Fe(H2O)5NO]^2 + is a complex formed during the brown ring test for NO^ - 3 ion. In this complex : Fe(H2O)5NO]^2 + is a complex formed during the brown ring test for NO^ - 3 ion. In this complex :](https://haygot.s3.amazonaws.com/questions/618410_582594_ans_1379fb9e77d54b43bda0e26d349c9fa9.png)






![Fe(H2O)5NO]^2 + is a complex formed during the brown ring test for NO^ - 3 ion. In this complex : Fe(H2O)5NO]^2 + is a complex formed during the brown ring test for NO^ - 3 ion. In this complex :](https://i.ytimg.com/vi/PIf9QoVJRFs/maxresdefault.jpg)