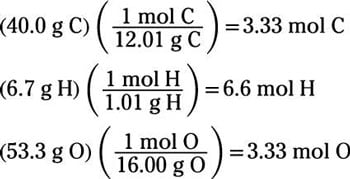If 3 moles of ethane (C2H6) are given, calculate the following:(i) Number of moles of carbon atoms. (ii) Number of moles of hydrogen atoms. (iii) Number of molecules of ethane.
How do we figure this out? We know that: 1) the number of oxygen atoms can be found by using Avogadro's number, if we know th

stoichiometry//moles: i understand you're looking for number or moles for hydrogen, but why do u multiply the number of hydrogen atoms by the amount of molecules in the compound?? i don't get


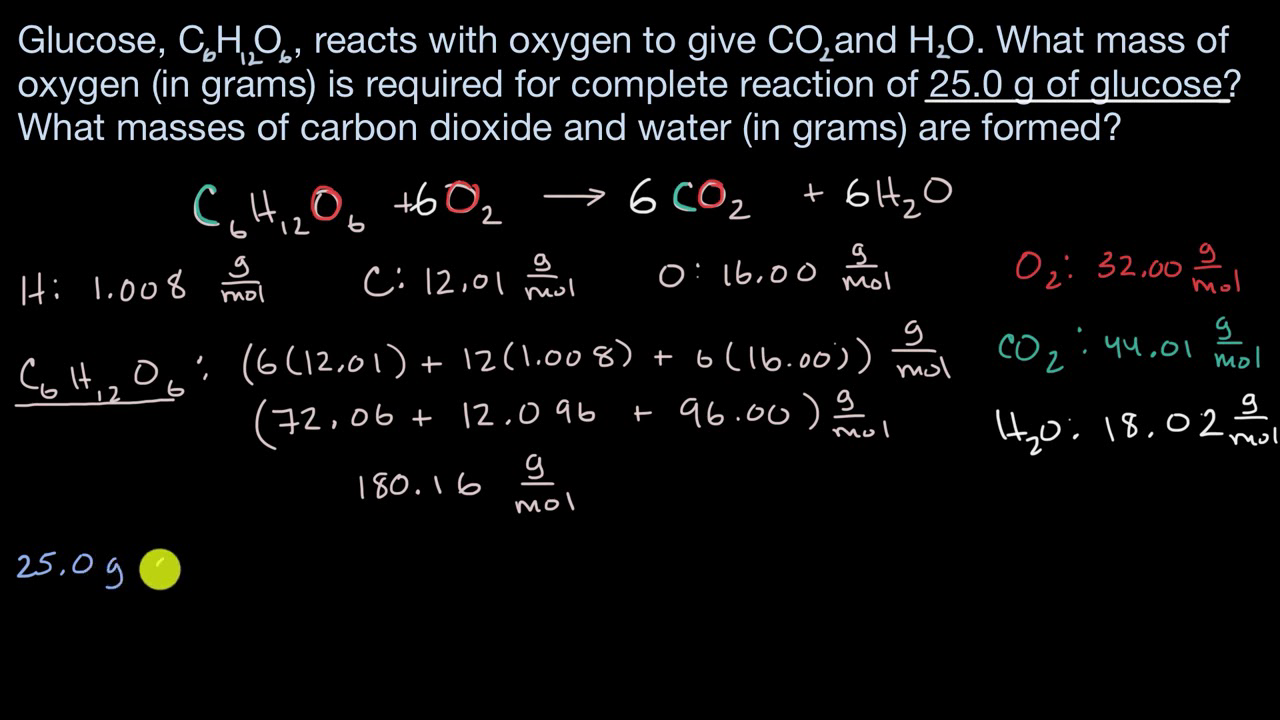
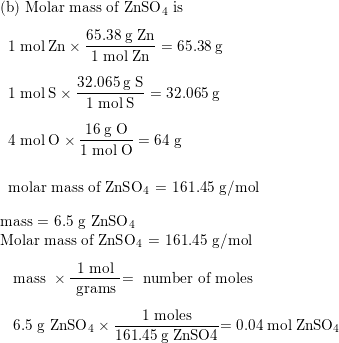


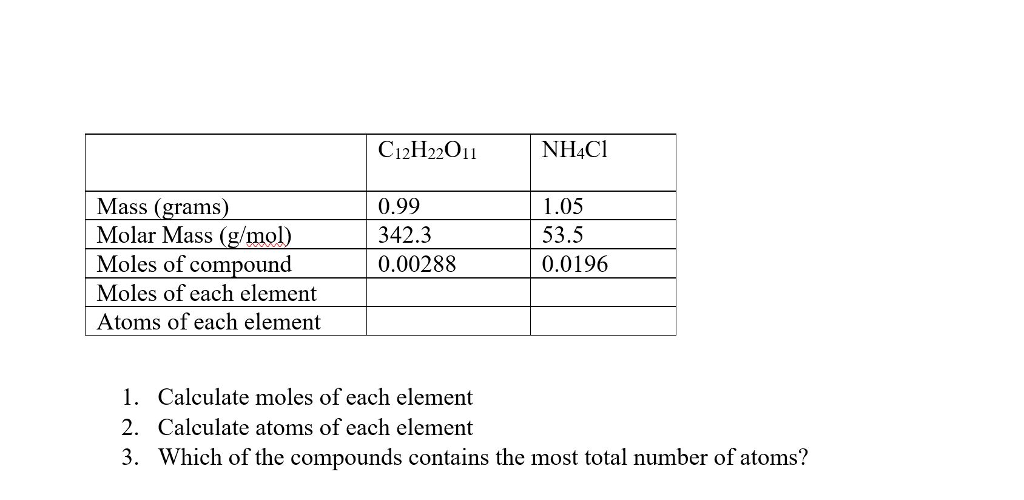
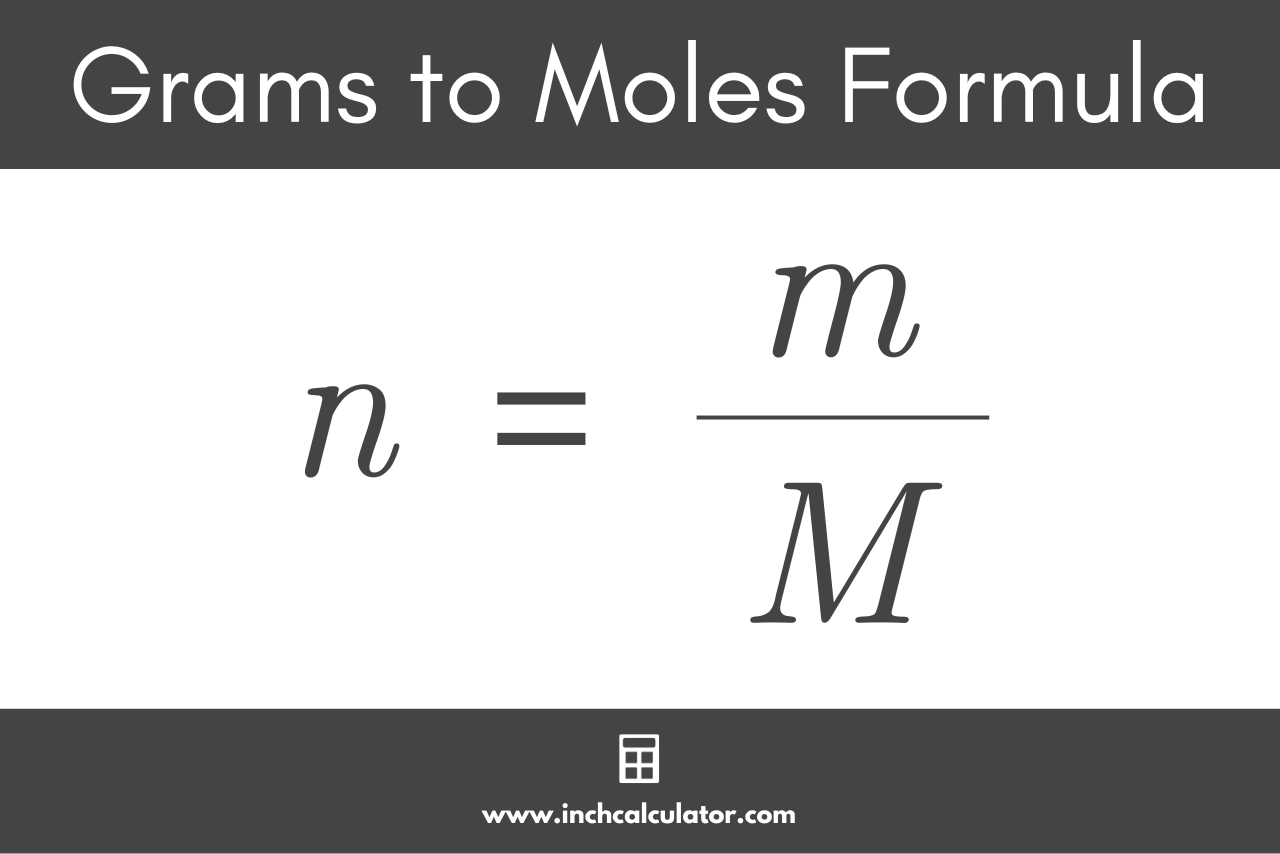
![Example] How to Calculate Moles From the Mass of a Compound - Converting Mass to Moles. - YouTube Example] How to Calculate Moles From the Mass of a Compound - Converting Mass to Moles. - YouTube](https://i.ytimg.com/vi/EKywe4xU4cs/maxresdefault.jpg)